TMA-6 synthesis
This is classical synthesis method of TMA-5 was criated with help of experts from BB forum.
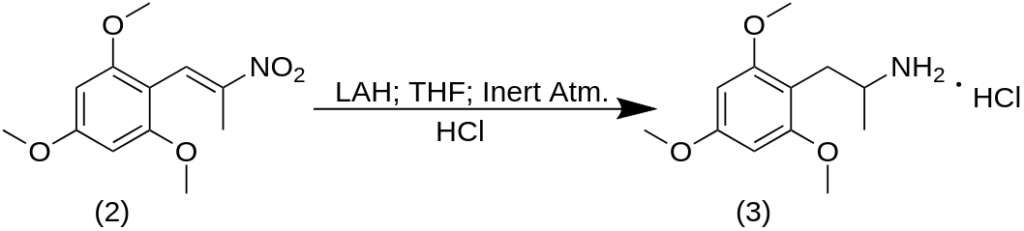
A solution of 5 g 2,4,6-trimethoxybenzaldehyde (1) in 20 g nitroethane was treated with 1.0 g of anhydrous ammonium acetate and held on the steam bath for 24 h. The excess solvent/reagent was stripped from the deep-red colored solution under vacuum yielding a residue that spontaneously set to a crystalline mass. This was well triturated under 5 mL MeOH, filtered, and washed with 3 mL additional MeOH to give 5.4 g of 2-nitro-1-(2,4,6-trimethoxyphenyl)propene (2) as yellow crystals. The m.p. of the crude material was 135-142 °C which could be raised to 147-148 °C by recrystallization from EtOH. The use of an alternate procedure for the synthesis of this nitrostyrene, using acetic acid as solvent and a stoichiometric amount of nitroethane (and ammonium acetate as catalyst), gave very poor yields. The use of butylamine as catalyst gave considerably better results.
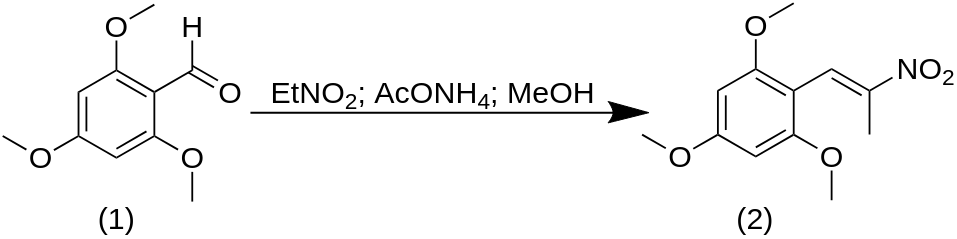
A suspension of 50 g LAH in 1 L anhydrous THF was placed under an inert atmosphere, stirred magnetically, and brought to a gentle reflux. There was added a total of 56.9 g 2-nitro-1-(2,4,6-trimethoxyphenyl)propene (2) as a saturated solution in THF. This was achieved by letting the condensed THF drip through a Soxhlet thimble containing the nitrostyrene with direct addition to the reaction mixture. The solubility was extremely low. The stirred mixture was maintained at reflux for 36 h, generating a smooth creamy gray color. After being brought to room temperature, the excess hydride was destroyed by the patient addition of 50 mL H2O, followed with 50 mL 15% NaOH (still some heat evolved) and then 150 mL additional H2O. Stirring was continued until the insoluble salts were white and loose. These solids were removed by filtration, and the filter cake washed with additional THF. The combined filtrate and washes were stripped of solvent under vacuum, and the 73 g of pale amber residue dissolved in 200 mL IPA, neutralized with approximately 50 mL concentrated HCl, and diluted with 2 L anhydrous Et2O. A lower, oily phase separated slowly set up as a crystalline mass. This was removed by filtration, Et2O washed, and allowed to air dry to constant weight. The weight of 2,4,6-trimethoxyamphetamine hydrochloride (3) was 41.3 g and the color was an off-white. There was a tendency to discolor upon air exposure. The m.p. was 204-205 °C which was increased to 207-208 °C upon recrystallization from IPA. The literature gives a m.p. of 214-215 °C for this salt after isolation and purification as the picrate salt (with a m.p. 212-213 °C from EtOH).