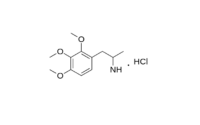
TMA synthesis
In this post I want to represent TMA (3,4,5-Trimethoxyamphetamine) synthesis from corresponding trimethoxy benzaldehyde 3,4,5-trimethoxybenzaldehyde. This is classical synthesis description was taken from BB forum.
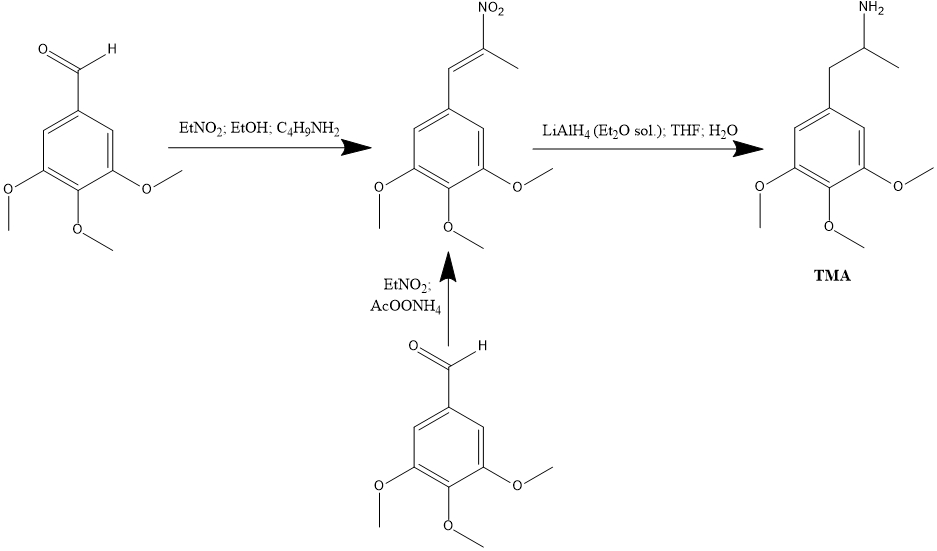
Procedure
To a solution of 39.2 g 3,4,5-trimethoxybenzaldehyde in 30 mL warm EtOH there was added 15.7 g nitroethane followed by 1.5 mL n-butylamine. The reaction mixture was allowed to stand at 40 °C for 7 days. With cooling and scratching, fine yellow needles were obtained which, after removal by filtration and air drying, weighed 48 g. Recrystallization from EtOH gave 2-nitro-1-(3,4,5-trimethoxyphenyl)propene as yellow crystals with a mp of 94-95 °C.
Alternatively, a solution of 20 g of 3,4,5-trimethoxybenzaldehyde in 75 mL nitroethane was treated with 4 g anhydrous ammonium acetate and heated on the steam bath until a deep red color had been generated. Removal of the excess solvent/reagent under vacuum gave a red oil which was dissolved in an equal volume of boiling MeOH. On cooling, yellow crystals of the nitropropene separated. Recrystallization from MeOH gave, after air drying to constant weight, 13.0 g with the same m.p.
Under an inert atmosphere, 38 g LAH was wetted with 100 mL anhydrous Et2O, and then suspended in 1 L dry THF. This was brought up to a gentle reflux, and there was added, slowly, a solution of 43.7 g 2-nitro-1-(3,4,5-trimethoxyphenyl)propene in 160 mL THF. Refluxing was continued for 36 h, and then the reaction mixture was cooled with an external ice bath. The excess hydride was destroyed by the cautious addition of 38 mL H2O, and this was followed by 38 mL 15% NaOH, and finally another 114 mL H2O. The inorganic salts which should have ended up as a loose, granular, easily filterable mass, looked rather like library paste, but they were filtered nonetheless. Washing with THF was attempted, but it was not efficient. The combined filtrate and washes were stripped of solvent under vacuum giving 31.5 g of the crude base as an amber oil. This was dissolved in 140 mL IPA, neutralized with concentrated HCl (15 mL was required), and diluted with 650 mL anhydrous Et2O. There was an initial oily phase which on continued stirring changed to pale pink solids. These were finely ground under CH3CN to give 15.2 g of 3,4,5-trimethoxyamphetamine hydrochloride (TMA) as white crystals that melted at 195-211 °C. All aluminum salts from everywhere were dissolved in dilute HCl, and 1 Kg of potassium sodium tartrate was added. There as added 25% NaOH allowed the pH to bring the pH to >9 without the precipitation of basic alumina. Extraction of this phase with CH2Cl2 was followed by removal of the solvent and salt formation as described above, allowed the isolation of an additional 6.4 g TMA. The product prepared in this manner contains some 10-15% 3,5-dimethoxy-4-hydroxyamphetamine as an impurity. A solution of 20 g of the TMA made in this manner in 200 mL 5% NaOH was extracted with 2×200 mL CH2Cl2. The pooled extracts were washed with 4×100 mL 5% NaOH, and the aqueous washes were pooled with the original base phase. The organic phase was stripped of its CH2Cl2 under vacuum to give an oil that was dissolved in 40 mL IPA, neutralized with concentrated HCl, and diluted with 400 mL anhydrous Et2O. There was the immediate formation of spectacular white crystals of pure 3,4,5-trimethoxyamphetamine hydrochloride, weighing 15.4 g and having a mp of 220-221 °C. The aqueous phase was brought to neutrality, treated with 10 g potassium di-hydrogen phosphate, brought to pH 9.0 with the careful addition of NaOH, and extracted with 5×100 mL CH2Cl2. Evaporation of the solvent under vacuum gave an oil that spontaneously crystallized. This product, 3,5-dimethoxy-4-hydroxyamphetamine could be further purified by sublimation at 130 °C at 0.2 mm/Hg. It was a white crystalline solid that slowly discolored in the air. The literature describes a picrate salt with a mp of 225 °C from EtOH.
Dosage: 100 – 250 mg
Duration: 6 – 8 h